Basic HTML Version
Chapter 2 — 2
Gases
exert pressure because they are made up of lots of fast moving molecules. The greater
the number and the faster they move, the greater the pressure.
Pressure on a Submerged Diver
The pressure acting on a submerged diver has two components:
1. The atmosphere above the water, termed
atmospheric pressure,
2. The weight of the water above the diver, termed
hydrostatic pressure.
Divers’ depth gauges
are calibrated only to read the hydrostatic pressure (the depth of water)
and so they read zero at sea level. They do not read the 1 atmosphere (1 ATA) above them.
Thus the “gauge pressure” is always 1 atmosphere less than the true or “absolute” pressure.
We will now elaborate.
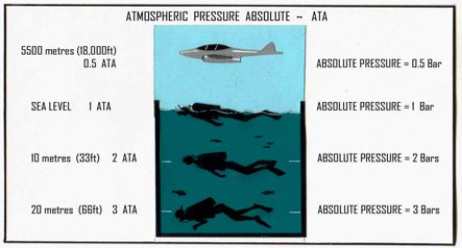
Atmospheric Pressure
The atmosphere above the earth is some 150 km high. Although air is very light, this amount
of air has significant weight and exerts substantial pressure on the earth's surface.
Atmospheric pressure at sea
level is referred to as "one
atmosphere" or "one bar". It
is the same as 101.3 kPa, 1
kg/cm
2
, 760mm Hg and 14.7
psi. At higher altitudes,
atmospheric pressure is
reduced, a factor which has a
significant effect on diving
in mountain lakes (see
Chapter 6).
Fig. 2.2 Atmospheric and Hydrostatic Pressures (depth)
added and thus converted to Absolute Pressure
ABSOLUTE
GAUGE
DEPTH
PRESSURE
PRESSURE
of SEAWATER
1 ATA
0 ATG
Surface
2 ATA
1 ATG
10 metres (33ft)
3 ATA
2 ATG
20 metres (66ft)
4 ATA
3 ATG
30 metres (99ft)
Table 2.1
Pressure at Depth

